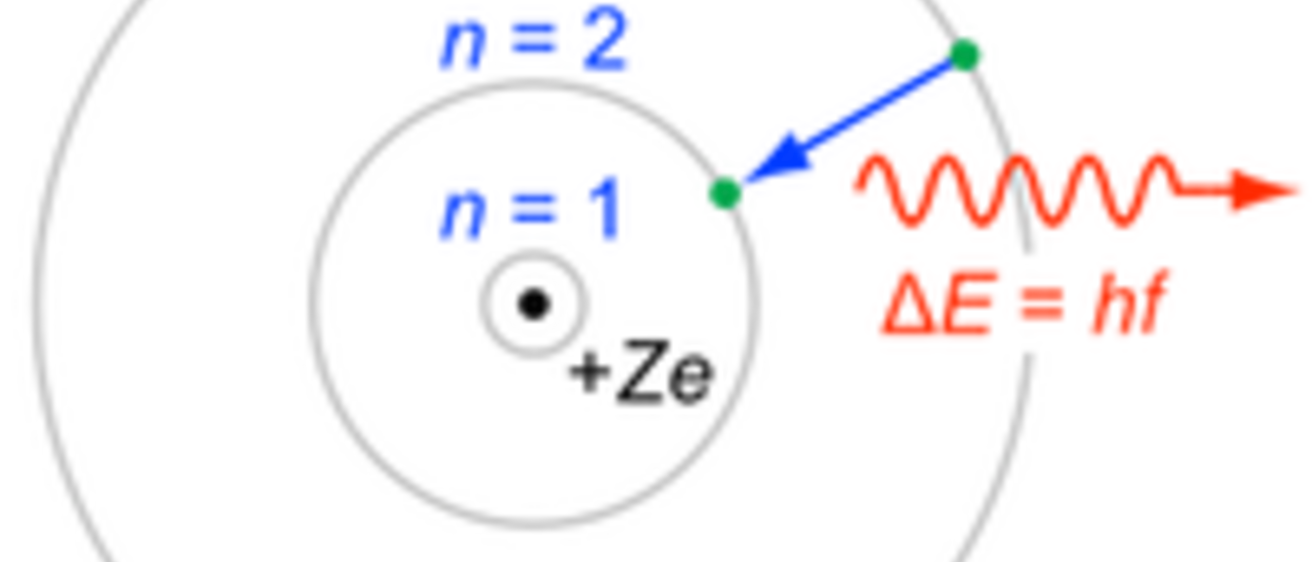
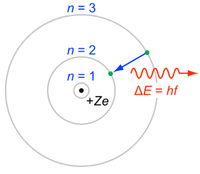
The spectra you have been observing is due to the gas being heated and are examples of emission line spectra. This process can also occur in reverse, with light of the line’s wavelength being captured by the atom in a process known as absorption. Absorption occurs anytime a cooler, thin gas is located in front of a white light source.
Your TA will have two light sources at the front of the room, a low pressure sodium lamp and a high pressure sodium lamp. You will view these lamps with your spectral glasses as well as with the ocean optics spectrometer.